|
Carbon-14 is constantlyĬonvert them into 14C atoms (the neutron is accepted and a proton is ejected from the nucleus). If 14C is constantly decaying, will the earth eventually run out of 14C? Which worldview does science support? Can carbon-14 dating help solve the mystery of which worldview is more accurate? The Bible teaches a young universe and earth. The secular (evolutionary) worldview interprets the universe and world to be billions of years old. The interpretation of past events is in question. The procedures used are not necessarily in question. Do all scientists accept the 14C dating method as reliable and accurate?Īll radiometric dating methods use scientific procedures in the present to interpret what has happened in the past.Are the dates provided by 14C dating consistent with what we observe?.Is the explanation of the data derived from empirical, observational science, or an interpretation of past events (historical science)?Īre there any assumptions involved in the dating method? Since the Bible is the inspired Word of God, we should examine the validity of the standard interpretation of 14C dating 
Genesis 1 defines the days of creation to be literal days (a number with the word “day” always means a normal day in the Old Testament, and the phrase “evening and morning” further defines the days as literal days). 
So we should never think it necessary to modify His Word. God knows just what He meant to say, and His understanding of science is infallible, whereas ours is fallible. When a scientist’s interpretation of data does not match the clear meaning of the text in the Bible, we should never reinterpret the Bible. In question, since 14C dates of tens of thousands of years are common. If this claim is true, the biblical account of a young earth (about 6,000 years) is (The electrons are so much lighter that they do not contribute significantly to the mass of an atom.) Carbon-14 DatingĬarbon-14 ( 14C), also referred to as radiocarbon, is claimed to be a reliableĭating method for determining the age of fossils up to 50,000 to 60,000 years. Atomic mass is a combination of the number of protons and neutrons in the nucleus. The atomic number corresponds to the number of protons in an atom. One is for potentially dating fossils (once-living things) using carbon-14 dating, and the other is for dating rocks and the age of the earth using uranium, potassium and other radioactive atoms. There are two main applications for radiometric dating. Some isotopes of certain elements are unstable they can spontaneously change into another kind of atom in a process called “radioactive decay.” Since this process presently happens at a known measured rate, scientists attempt to use it like a “clock” to tell how long ago a rock or fossil formed. The illustration below shows the three isotopes of carbon. An “isotope” is any of several different forms of an element, each having different numbers of neutrons. 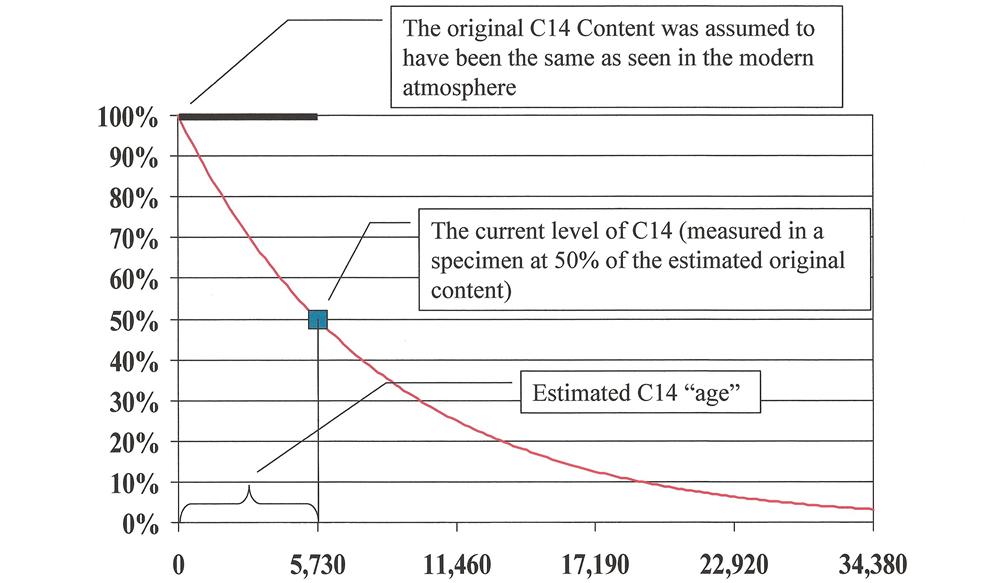
So, a carbon atom might have six neutrons, or seven, or possibly eight-but it would always have six protons. 
The number of neutrons in the nucleus can vary in any given type of atom. For example, all carbon atoms have 6 protons, all atoms of nitrogen have 7 protons, and all oxygen atoms have 8 protons. The number of protons in the nucleus of an atom determines the element. Protons and neutrons make up the center (nucleus) of the atom, and electrons form shells around the nucleus. Atoms are made up of much smaller particles called protons, neutrons, and electrons. Recall that atoms are the basic building blocks of matter. Before we get into the details of how radiometric dating methods are used, we need to review some preliminary concepts from chemistry.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
